
On the nature of pain
Last week, a manatee was found in Florida waters, with the word ‘Trump’ scraped on its back. Although this kind of ruthless mutilation is horrific in itself, I started wondering if the animal felt any kind of pain.
I must admit, up until I came across the news about what happened to the manatee in Florida, I knew very little about manatees, in general. And my first thought was whether, during the scraping proccess, this manatee suffered at all. To my dismay, there were not many scientific papers dealing with the somatosensory system in manatees. However, I did find something that eased my soul a little bit: one of the articles reporting on the dreadful event states that the scratch was done in the algae growing on the animal’s back. Still, in the same article it is said that manatees have sensory hairs and nerves in their skin, which means that, if the cuts had touched the skin, they could have caused pain; not to mention the infection that the skin was at risk of, due to the open wounds.
The video below shows the above-mentioned manatee swimming, with the human-made scars on its back.
After reading all these news articles, I was left with some questions that kept occupying my mind: What are manatees?; To what extent can they feel pain?; And can we talk about ‘pain’ at all in manatees, or just nociception? Lastly, how did pain evolve throughout the animal kingdom?
What are mantees?
Also known as ‘sea cows’, manatees (Trichechus manatus latirostris) are herbivorous acquatic mammals of the Order Sirenia. As the name of their order suggests, manatees are believed to be the animals behind the myths of mermaids. For those interested in how manatees inspired mermaid legends, please check out the video below.
Manatees are the largest vegetarian animal to inhabit the sea, and they communicate with each other through high-pitched sounds. They are also very gentle and lack defense mechanisms, given that they do not have any natural enemies. However, they have become and endangered species, due to human activity, which is the manatee’s greatest threat. According to the Florida Fish and Wildlife Conservation Commission, the year 2020 was a hard one for manetees as well: 637 of them died, 90 of which were victims of boat collisions, and another 15 were killed by other interactions with humans.
Although manatees do not possess a highly acute visual system, they compensate for that by the presence of tactile hairs, or vibrissae, spread all over their body, especially on the face. This distribution of vibrissae is something unique among mammals, and to manatees it is highly useful in allowing them to navigate in the water.
Since mantees rely tremendously on tactile inputs, it comes as no surprise that their brains are organised to support somatosensation. The primary somatosensory cortex of manatees occupies 25% of their neocortex. Moreover, the sixth layer of their cortex contains clusters of neurons, known as Rindenkerne, which are believed to process information related to the manatee’s facial and bodily vibrissae. Although the Rindenkerne cells of manatees are somewhat similar to other cortical representations of vibrissae, termed ‘barrels’, in rodents, shrews, opposums and hedgehogs, Rindenkerne are unique to sirenia. These neuronal aggregates become active when manatees engage in tactile exploration and object recognition.
At the subcortical level, manatees possess three types of somatosenroy nuclei in their brainstems, namely the Birchoff’s nucleus, which receives information from flukes, the cuneate-gracile nucleus, which processes inputs from flippers and body trunk, and the trigeminal nucleus, which receives sensory inputs from facial vibrissae. Figure 1. below shows the somatosentory representations of the manatee’s body parts, in a coronal section of the brainstem. The thalamus also has specialised somatosensory nuclei, which differ in size, depending on their functional relevance to somatic sensation.


Figure 1. Left diagram based on image by Isuru Pryiaranga. Right image from Sarko et al. (2007), showing functional divisions withing the brainstem, corresponding to the manetee’s body parts.
Given that somatosensation is so developed in manatees, one burning question is whether they feel pain.
What is pain?
Pain is different from nociception. However, pain from injury cannot occur without nociception. The latter reffers to the process of detecting injury by the activation of a special class of receptors found in the skin, as well as deep tissues and organs, known as nociceptors. The detection of potentially or actually damaging stimuli is followed by a reflex withdrawal reaction, or nociceptive behaviour, mediated by nerves in the spinal cord. The nerve fibres that detect noxious stimuli are Aδ fibres and C fibres, which have their cell bodies in the dorsal root ganglion (DRG) of the spinal cord, as shown in Figure 2.
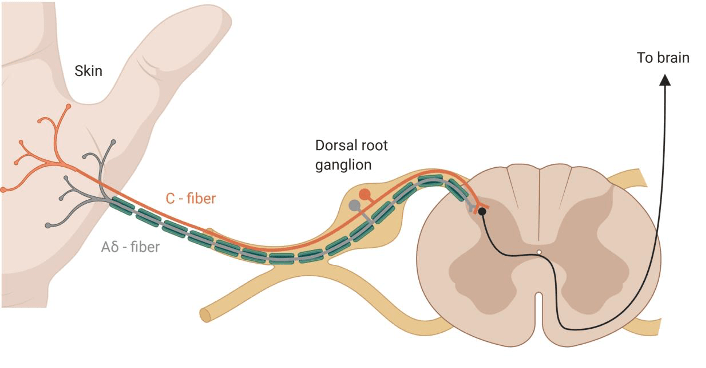
Aδ fibres are mechano-nociceptors, meaning that they are activated by high mechanical pressures. C fibres are polymodal, which means that they respond to a variety of noxious stimulations, such as noxious chemicals (e.g., acids), extreme temperatures and high mechanical pressures. They not only encode the stimulus modality (type), but also their intensity and duration, which are relayed to reflex centres in the central nervous sytem, mediating withdrawal reactions.
The nociceptive information travels from the DRG to different parts of the brain via spinothalamic tracts (from the spinal cord to the thalamus) and sensory fibres of the trigeminal tract (from the face to the thalamus). And it is within the brain that pain happens.
Pain is a complex feeling. Many brain areas are involved in not just generating pain, but also in ameliorating it. Structures from the limbic system, such as the amygdala, receive and integrate nociceptive and affect-related information. The amygdala can lead to increased nocifensive and affective pain behavior, while, under certain circumstances, it can also contribute to endogenous pain inhibition. Pain is also processed in the hypothalamus, the basal ganglia, the insula and the somatosensory cortices. Because these areas play a role in metabolism, as well as fear, pleasure and homeostasis, the nociceptive information is integrated and modulated according to the current state of the individual. In some situations, pain becomes pathological, as it is the case in neuropathic pain, where either previously innocuous stimuli become painful (aka, allodynia), or previously painful stimuli become even more painful (aka, hyperalgesia).
There are two brainstem structures, which are highly involved in controlling pain and generating analgesia. One of them is the periaqueductal grey (PAG) and the other is the rostral ventromedial medulla. These regions exert control over pain to prioritise competing stimuli, and to maintain homeostasis and survival. You might have noticed that, in highly stressful situations you do not feel pain. This is known as stress-induced analgesia, a phenomenon whereby the brain responds to stress by the production of endogenous opioids that act as natural analgesics in the nervous system. The opioid receptors found in the brain are the same ones which analgesic drugs, such as synthetic opioids and morphine, act on to relieve pain.
The evolution of pain
Many animal taxa have nociceptors. A schematic of the evolutionary development of nociceptors and the types of noxious stimuli they respond to is presented in Figure 3. In order to process nociceptive inputs, animals need a central nervous system (spinal cord and brain). It might come as a surprise that such a system, though at different levels of complexity, is found in all kinds of animals, including insects (like Drosophila melanogaster, the fruit fly), C. elegans (a type of worm highly studied in neurosciences), fish, amphibians, reptiles, birds and, of course, mammals.

Taken from Sneddon (2017)
Life-history shapes pain perception. A very interesting example is the African naked mole rat, which lives in underground burrows that are poorly ventilated, hence contain high carbon dioxide levels. As a result, the C fibres of the naked mole rat are unresponsive to acid, which means that, while other mammals find acidic environments nociceptive, the African naked mole rat does not.
When it comes to acquatic animals, such as manatees, they are expected to have differences in their sensory system compared to terrestrial ones, due to distinct ecological and evolutionary pressures. In water, any chemicals become dilluted, shifts in temperatures are less common, and there is no mechanical damage due to falling. Thus, acquatic animals are possibly at a lower risk of damage than terrestrial animals, which has implications on their nociceptive system.
As far as manatees go, it is still unclear to what extent they feel pain. The fact that they are an endangered species makes is difficult to study them. But given that they posses a very well-developed somatosensory system, which is even more advanced than in other mammals, it is expected that manatees are familiar with pain. Moreover, we still do not know enough about their stress, fear, memory and pleasure systems, which all play a role in pain processing.
It would be great if we managed to achieve a better understanding of these amazing marine animals. But, until then, let us enjoy their existance peacefully, without interfering violently with their lifestyles and without exposing them to any potential pains.
For a more in-depth view on pain, as well as more information about manatees, I highly encourage you to read the papers and articles listed in References.
Special thanks to Isuru Priyaranga for creating the cover image. He is a fellow blogger and YouTuber, and I highly recommend visiting his blog and YouTube Channel.
References
- Neugebauer, V. (2007). Amygdala—Pain Processing and Pain Modulation. In: Zhuo M. (eds) Molecular Pain. Springer, New York, NY. doi:org/10.1007/978-0-387-75269-3_20
- Sarko, D.K., Johnson, J.I., Switzer, R.C., III, Welker, W.I. and Reep, R.L. (2007). Somatosensory Nuclei of the Manatee Brainstem and Thalamus. Anat Rec, 290:1138-1165. doi: org/10.1002/ar.20573
- Sneddon, L.U. (2017). Comparative Physiology of Nociception and Pain. Physiology, 33:63-73. doi:10.1152/physiol.00022.2017
- How did manatees inspire mermaid legends?
- A Manatee Was Found With ‘Trump’ Scraped Into Its Back
- Florida manatee with ‘Trump’ etched on back prompts investigation
- Manatees amid vegetation









